Calorimetry and CPET — Measuring Energy Through Breath
Every breath we take during exercise carries a clue about how efficiently our body converts fuel into energy. Calorimetry is the science that helps us measure this energy expenditure — and when combined with Cardiopulmonary Exercise Testing (CPET), it becomes one of the most insightful tools in functional physiology.
What Is Calorimetry in Simple Terms?
Calorimetry helps us understand how the body produces energy — whether at rest or during exercise.
In CPET, this is done indirectly, by measuring how much oxygen (O₂) is taken in and how much carbon dioxide (CO₂) is released during breathing.
That’s why modern CPET systems are called indirect calorimeters — they don’t capture heat directly, but instead calculate it from the gases we inhale and exhale.
The Basic Biochemistry & Fuel Use in Calorimetry
The basic biochemical process can be summarised as:
Foodstuffs + O₂ → Heat + CO₂ + H₂O
This simply means that when oxygen reacts with nutrients, energy (heat) is released and carbon dioxide and water are produced as by-products.
In indirect calorimetry, we don’t measure heat directly. Instead, oxygen consumption (V̇O₂) and carbon dioxide production (V̇CO₂) are measured at the mouth. These gas-exchange values are then used to calculate the metabolic energy expenditure — making it a practical alternative to direct calorimetry during exercise testing.
Representative Oxidation Reactions for Different Fuels
Carbohydrates (Glucose)
C₆H₁₂O₆ + 6 O₂ → 6 CO₂ + 6 H₂O + energy
Fats (Palmitic Acid)
C₁₆H₃₂O₂ + 23 O₂ → 16 CO₂ + 16 H₂O + energy
These reactions show that both carbohydrates and fats require oxygen for oxidation — but in different proportions. That’s exactly why changes in V̇O₂ and V̇CO₂ during CPET reveal which fuel the body is using at any given workload.
Anaerobic Contribution
In normal physiological conditions, the human body relies mainly on aerobic metabolism — oxygen-driven energy production that keeps the system efficient during everyday activity and steady-state exercise. Glucose is broken down to pyruvate through glycolysis and then fully oxidized to CO₂ and H₂O when oxygen is available.
However, during short bursts of high-intensity effort — such as a 100-meter sprint — oxygen delivery cannot keep up with the rapid ATP demand. When this happens:
- Pyruvate cannot enter aerobic oxidation fast enough
- It is diverted toward anaerobic pathways
- Lactate becomes the end product
This is the body’s temporary backup to generate ATP quickly when oxygen supply is insufficient.
During recovery, the accumulated lactate is gradually converted back to glucose in the liver through gluconeogenesis. This process requires additional oxygen — creating what we call oxygen debt.
That extra oxygen consumed after intense effort reflects:
- Lactate clearance
- Restoration of muscle and liver glycogen
- Recovery of metabolic equilibrium
In a CPET context, this shift between aerobic and anaerobic metabolism can be clearly seen through rising V̇CO₂, RER approaching or exceeding 1.0, increased ventilation, and the characteristic gas-exchange patterns.
Open-Circuit Indirect Calorimetry Techniques in CPET
Modern CPET systems use the open-circuit indirect calorimetry technique. (Refer figure 01 Indirect calorimetry)
In this method, the subject breathes room air, and the system continuously measures:
• Inspired and expired air volumes (V̇I and V̇E)
• Oxygen and carbon dioxide fractions (FIO₂, FEO₂, FICO₂, FECO₂)
From these, the metabolic module computes:
V̇O2 = V̇IFIO2 - V̇EFEO2
and
V̇CO2 = V̇EFECO2 - V̇IFICO2
These equations express oxygen uptake and carbon dioxide output in real time — the core of how energy expenditure is derived during CPET.
Because air flows freely (open to the atmosphere), this is termed an open-circuit system. It allows the subject to breathe naturally and respond to increasing workload, making it ideal for exercise studies.
By contrast, closed-circuit calorimetry involves breathing from a sealed reservoir with a fixed gas mixture (often pure O₂). It’s mainly used for laboratory or resting metabolic research because it limits natural ventilation.
All modern CPET systems use open-circuit indirect calorimetry, ensuring accurate breath-by-breath measurement of VO₂ and VCO₂ while the subject breathes freely.
The Science Behind the Calculation
The principle behind indirect calorimetry is summarized in the modified Weir Equation:
Energy Expenditure (kcal/min) = 3.941 × VO₂ + 1.106 × VCO₂
This equation converts respiratory gas data into energy units. It is based on the known caloric value of oxygen when oxidizing carbohydrates and fats. During CPET, this calculation runs continuously—each breath is analyzed in real time, allowing the system to compute energy expenditure as workload changes. It can also determine Resting Energy Expenditure (REE) when no external workload is applied.
RER — The Window into Substrate Use
The Respiratory Exchange Ratio (RER) describes the relationship between oxygen uptake and carbon-dioxide output:
RER = VCO₂ / VO₂
RER provides insight into which substrate the body is oxidizing to meet energy demand.
At Rest or Low Workloads — Fat Dominance (RER ≈ 0.7)
During low-intensity activity, oxygen is plentiful and energy comes mostly from fat oxidation:
C₁₆H₃₂O₂ + 23 O₂ → 16 CO₂ + 16 H₂O
RER = 16 / 23 = 0.70
Moderate to High Workloads — Carbohydrate Dominance (RER ≈ 1.0)
As intensity increases, ATP needs rise quickly, and carbohydrates become the preferred fuel:
C₆H₁₂O₆ + 6 O₂ → 6 CO₂ + 6 H₂O
RER = 6 / 6 = 1.0
Very High Workloads — Anaerobic Contribution (RER > 1.1)
Beyond the ventilatory threshold, excess CO₂ is produced from lactic-acid buffering:
H⁺ + HCO₃⁻ → H₂CO₃ → CO₂ + H₂O
This elevates RER above 1.0 and signals near-maximal exertion.
Fuel Utilization Shift with Exercise Intensity
As aerobic power increases, the balance between fat and carbohydrate use shifts progressively. At rest, approximately 60–70% of energy may come from fat. With rising workload, carbohydrate contribution increases while fat oxidation declines.
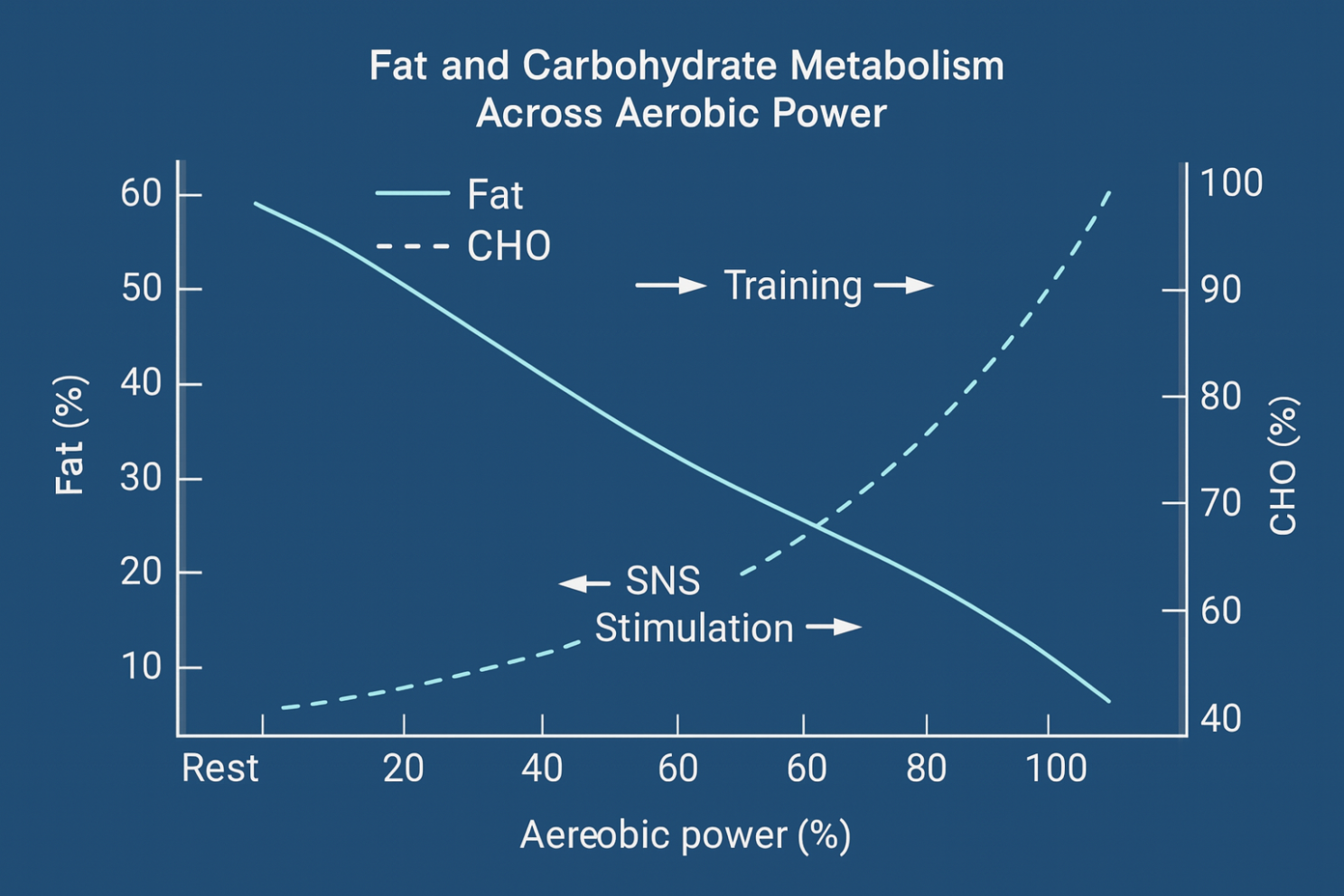
Figure 02. Fat and Carbohydrate Metabolism across aerobic power.
This metabolic crossover happens because:
• Sympathetic activation speeds glycogen breakdown and carbohydrate oxidation.
• Oxygen availability per ATP demand decreases at higher intensity, favoring glucose.
• Training adaptation can delay this crossover, improving fat-use efficiency at moderate intensities.
Why Calorimetry Matters in CPET
Calorimetry adds another layer to CPET beyond oxygen uptake and ventilation. It helps clinicians understand how efficiently the body converts fuel into work.
Some key insights it provides:
- Energy expenditure — quantifies total metabolic cost of exercise.
- Metabolic efficiency — compares oxygen use versus work output.
- Substrate utilisation — shows the shift between fat and carbohydrate metabolism.
Clinically, this information helps in:
- Heart failure — assessing metabolic inefficiency and reduced oxygen utilisation.
- COPD — measuring the oxygen cost of breathing.
- Obesity and malnutrition — determining accurate caloric requirements and rehabilitation goals.
In simple terms, calorimetry in CPET helps you see not only how much a person can perform, but how efficiently their body is working while doing it.
Technology Behind Modern CPET Systems
Modern CPET systems integrate advanced sensing technologies to achieve precision and long-term reliability.
They use:
- Pneumotach and ultrasonic flow sensors for precise, linear ventilation measurement.
- Paramagnetic and galvanic O₂ cells for accurate measurement of inspired/expired oxygen.
- NDIR and ultrasound-based CO₂ analyzers for stable, drift-free carbon-dioxide measurement.
These technologies ensure breath-by-breath accuracy, minimal drift, and high reproducibility, which are critical when measuring VO₂, VCO₂, and RER.
The combination of these sensors allows modern systems to capture rapid physiological changes with precision — ensuring even transient ventilatory and metabolic responses are recorded in real time.
Summary
Calorimetry bridges metabolism and physiology. Within CPET, it translates breath-by-breath gas exchange into real energy data — helping us understand how fuel is used, how efficiently energy is produced, and how the body responds under increasing stress.
It’s not just about performance; it’s about efficiency, adaptation, and clinical insight.
Frequently Asked Questions (FAQ)
Calorimetry & CPET
- What does indirect calorimetry measure in CPET?
VO₂ and VCO₂ — breath-by-breath. - Why not use direct calorimetry in CPET?
It requires a sealed heat chamber — not practical during exercise. - Is RER the same as RQ?
No. RQ is cellular; RER is respiratory. - Why does RER exceed 1.0?
Because of lactic-acid buffering producing excess CO₂. - What is normal resting RER?
0.75–0.85. - What does the Weir equation do?
Converts VO₂ and VCO₂ to kcal/min or kcal/day. - Can CPET measure REE?
Yes — from a stable resting phase. - Do fats and carbohydrates require different O₂?
Yes — reflected in RER behavior. - What influences energy expenditure?
Workload, training status, oxygen delivery, ventilation, movement economy. - Are VO₂/VCO₂ measurements accurate?
Yes, with proper sensor calibration and delay correction. - Can CPET identify limiting systems?
Yes — cardiac, pulmonary, muscular, or deconditioning. - What RER indicates peak effort?
RER ≥ 1.05 to 1.10. - Do manufacturers use different sensors?
Yes, but all follow the same physiological equations. - Which is more important — VO₂ or RER?
Both. VO₂ reflects capacity; RER reflects metabolic strategy.
📖 References
- Wasserman K., et al. Principles of Exercise Testing and Interpretation.
- Weir J.B. Journal of Physiology, 1949.
- Gaesser & Brooks. Med Sci Sports Exerc.
- Brooks & Fahey – Exercise Physiology
- Jones & Campbell. Clinical Exercise Testing.
- Péronnet & Massicotte – Substrate Utilisation Tables
- Lehninger - Principles of Biochemistry, Seventh Edition
